A clinical grade stem cell culture for regenerative medicine, StemFit Basic03, has been launched by Amsbio.
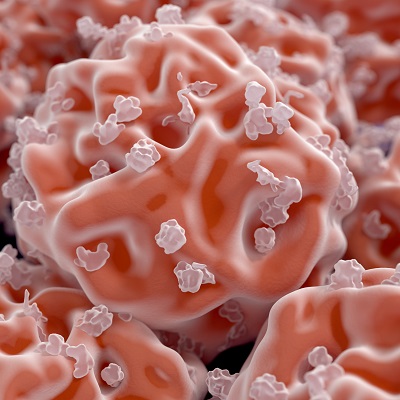
StemFit cell culture medium is formulated for pluripotent stem cells, says Amsbio
Provided ready to use from an animal-origin free, the StemFit Basic03 medium provides stable and reproducible culture condition for induced pluripotent stem and embryonic stem cells, says the company.
A Japanese version, StemFit AK03N, has been accepted by the Japanese Pharmaceuticals and Medical Devices Agency (PMDA) for retinal regenerative medicine clinical trials using human induced pluripotent stem cells (hiPSCs).
The product is free of animal- and human-derived components, and combines high colony forming efficiency with low media volume consumption for cost-effective colony expansion.
In an independent study, media of different formulations for culturing iPS cells were compared.
The research showed that iPS cells cultured with StemFit Basic03 demonstrated higher cell proliferation, lower lactate accumulation, and better homogeneity of gene expression, than cells cultured with four other media.
A poster describing this study is available at www.amsbio.com/presentation/stemfit-catapult-poster.pdf
